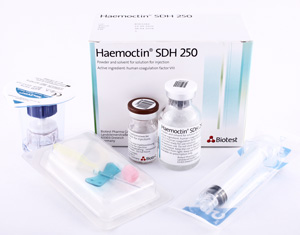
Haemoctin® SDH is a chromatographically purified human coagulation factor VIII preparation with a physiological concentration of von Willebrand factor (VWF), which is marketed as a freeze-dried powder in presentations of 250 IU, 500 IU and 1000 IU of FVIII per vial. The final product comprises two vials (one for the powder and one for the solvent sWFI), a disposable syringe (5 ml or 10 ml), a transfer set with integral filter and a butterfly needle.
Haemoctin® SDH is produced from human plasma. After reconstitution with sWFI Haemoctin® SDH 250 and Haemoctin® SDH 500 contain approximately 50 IU/ml human coagulation FVIII, whereas Haemoctin® SDH 1000 contains approximately 100 IU/ml human coagulation FVIII. The specific activity of Haemoctin® SDH 250, 500 and 1000 is about 100 IU FVIII/mg protein. In addition, Haemoctin® SDH contains glycine, sodium chloride, sodium citrate, calcium chloride dihydrate as excipients.
Additional Information
Haemoctin® SDH is licensed
- for the prophylaxis and treatment of bleedings in patients with haemophilia A,
- for the treatment of acquired factor VIII deficiency and
- for the treatment of patients with factor VIII inhibitors.
Although Haemoctin® SDH contains defined quantities of von Willebrand factor (VWF), it is not licensed for the treatment of von Willebrand’s disease. VWF performs numerous functions in haemostasis. One important function is the stabilization of the factor VIII structure. Without VWF, factor VIII would have only a short lifetime in the circulation.
In addition to the prophylaxis and treatment of bleeding, factor VIII is also employed in various immunotolerance therapy (ITT) schemes, in both congenital and acquired inhibitor haemophilia.
Please refer to the full prescribing information for more details.